Pep talk - December 2025

Commercially available media – the hidden pitfalls of proprietary formulations
Liz Stewart, Senior Product Manager at Qkine gives her view on the problems with proprietary media formulations.
Currently, the trend is shifting towards fully proprietary supplements tailored to specific stem cell and organoid types. These can be appealing, particularly for labs starting out with a new stem cell or organoid type, as they are a quick and pre-tested solution which reduces the need for lengthy optimization. This is particularly valuable when working with precious patient samples, where time-consuming protocol development isn’t feasible. Increasingly, however, we hear regrets from labs that have relied on such proprietary media formulations.
Reliance on proprietary media supply can lead to a host of issues down the line:
- What is in it? Formulations with no transparency in contents and no recipe, give no scope for refinement or optimization.
- What is the source? Commercial media formulations often contain multiple growth factors, but very few companies manufacture these proteins. So where do the growth factors in these formulations come from? How pure are they?
- Supply and cost. Supply issues when relying on a specific reagent from a single source, particularly if it is short dated can lead to projects being put on hold, and costs can make scale-up expensive or impossible.
For researchers looking to move away from proprietary formulations to enable protocol refinement and scalability, the question is: when? There is rarely a good time, especially when resources are limited, but the sooner the switch is made, the less painful it will be in the long run.


Refined and benchmarked homemade media for cost-effective, weekend-free human pluripotent stem cell culture
In a recently revised and expanded paper from our collaborators in the Bertero lab, Truszkowski et al. have described a cost-effective homemade media recipe for the culture of human pluripotent stem cells (hPSCs).
Along with our recent application note - Commercial versus in-house media: a comparative study of human induced pluripotent stem cell maintenance, this adds to the evidence that homemade media using Qkine growth factors can be a suitable replacement for, or improvement on, commercially available media.
The high cost of commercially available proprietary media and lack of transparency and flexibility can be a problem for academic labs and translational labs looking at scaling iPSC protocols.
Application note: Commercial versus in-house media: A comparative study of human induced pluripotent stem cell maintenance
Since their initial description, human iPSCs have transformed biomedical research by enabling patient-specific disease modeling and providing a renewable source of cells for drug screening and regenerative therapies. The maintenance of iPSCs in an undifferentiated and genetically stable state demands culture conditions that accurately replicate essential signaling environments while minimizing the use of undefined components. Early feeder-dependent systems relying on mouse embryonic fibroblasts have largely been replaced by feeder-free, chemically defined media, reducing variability and enhancing clinical applicability.
This application note directly compares the performance of selected commercially available iPSC media with in-house formulations. Key parameters assessed include cell morphology and proliferation rate. By systematically evaluating both types of media, we aim to provide practical insights for laboratories seeking to balance cost, performance, and experimental reproducibility in human iPSC culture systems.
| Media Type | Cost per 500 ml |
| Essential 8 (E8) | £268.00 |
| TeSR E8 | £248.00 |
| NutriStem® hPSC XF GF-free + Qkine FGF2-G3 and TGF-β1 | £259.20 |
| E8-like media + Qkine FGF2-G3 and TGF-β1 | £110.12 |
Cost per 500 ml for commonly used iPSC media, correct as of August 2025.
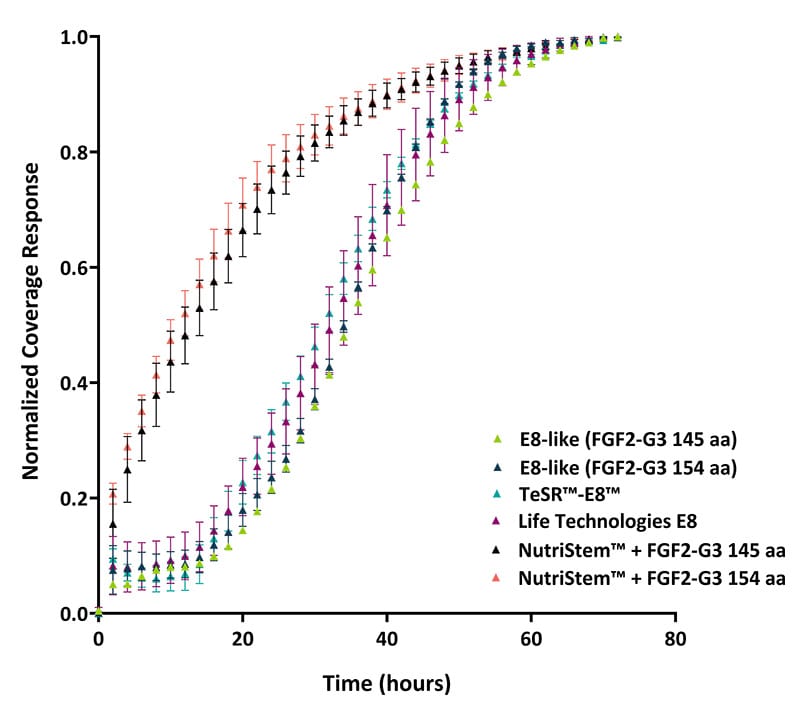
Comparison between Qkine defined homemade media preparations (E8-like) and proprietary media preparations for iPSC maintenance. Quantified using Axion Omni live-cell imaging system.
Pricing update January 2026
At Qkine, we’re committed to providing the high-quality and high purity growth factors & cytokines you have come to rely on for your stem cell culture applications. To help us continue delivering the standards you expect, our list prices will be updated from 1st January 2026. Read more
Last shipping dates of 2025 & first shipping dates of 2026
View our last shipping dates for 2025, dates for some areas can be extended if customers can guarantee they can take delivery up to 23rd December. Read more
Did you miss?
Raising the Standard: Developing a High-Quality IL-34 Protein
Despite its importance, scientists have long struggled to access a reliable, animal-free, and high-quality version of IL-34. Most available proteins on the market either don’t work well or vary too much from batch to batch. This makes experiments harder to reproduce and wastes both time and resources, something researchers simply can’t afford.
When we first started working on IL-34 at Qkine, we weren’t just trying to make another recombinant protein. We wanted to solve a real problem. We knew that if we could develop an IL-34 that was consistent, highly active, and free from animal-derived components, it could have a big impact on research.
Qkine launches cell therapy grade recombinant proteins to support next-generation therapies
Cell therapy is rapidly transforming modern medicine, offering new hope for patients with cancer, autoimmune conditions, and genetic disorders. However, the development and manufacturing of these therapies demand reagents of uncompromising quality, consistency, and safety. Qkine’s new Cell Therapy Grade proteins address this critical need.
Why are Qkine’s new products labelled cell therapy grade and not GMP grade?
Qkine have recently released a range of growth factors and cytokines specifically designed to be suitable for cell and gene therapy manufacturing. Most recombinant protein manufacturers would describe these as 'GMP grade' but we haven't, so why are the products labelled cell therapy grade and not GMP grade? Our latest blog explains what exactly is GMP and how our cell therapy grade proteins comply with GMP and ISO standards.
Animal origin-free protein manufacture for cell therapy
We have previously discussed the issues surrounding animal-derived products for research and why animal origin-free (AOF) alternatives are preferred. These include the potential for cross-contamination with other bioactive proteins or contaminants, which can introduce unwanted variability. When considering proteins used for the development of cell therapy, further assurance of purity and consistency is paramount, particularly due to the potential risk of harm through the application of an improperly manufactured product.
New differentiation kits
At Qkine we believe in transparency, but we also believe in convenience and saving money! Our differentiation kits contain everything you need for complex differentiations of PSC into mature cell types. No secrecy, just 30-40% saving on individual list prices.
Paired with our extensive range of application notes using the kits for full knowledge sharing and validation.
We're always happy to collaborate so if you think something is missing, let us know!
Microglial differentiation kit (Qk519)

Human BMP-4 - Qk038 - 25 µg
Used for differentiation of mesenchymal cells to adipocytes, epithelial cancer EMT, and regulating neuronal and glial cell development.
FGF2-G3 145 aa - Qk052 - 50 µg
A thermostable engineered form of FGF-2. FGF2-G3 145 aa is the 145 aa active domain of FGF-2 with the functional half-life increased from <10 h (wild-type) to >7 days (FGF2-G3).
Human Flt3L - Qk087 - 25 µg
Stimulates the survival, proliferation, and differentiation of various early myeloid and lymphoid progenitor cells. Flt3L is commonly used in the differentiation of hematopoietic stem cells into dendritic cells.
Human GM-CSF - Qk076 - 25 µg
Commonly used in cell culture to stimulate the differentiation and maturation of human-induced pluripotent stem cells or peripheral blood monocytes to myeloid cells.
Human M-CSF - Qk075 - 25 µg
Used in vitro for the differentiation of human induced pluripotent stem cell (iPSC)-derived macrophages as well as the maintenance and development of monocytes in hematopoietic stem cell culture.
Human IL-3 – Qk090 - 25 µg
Stimulate the differentiation and maturation of human induced pluripotent stem cells towards mast cells, basophils, neutrophils, eosinophils, monocytes, and megakaryocytes.
Human IL-34 PLUS™ – Qk091 - 25 µg
Plays a key role for the development and maintenance of tissue-resident macrophages such as Langerhans cells in the skin and microglia in the brain.
Human SCF - Qk078 - 25 µg
Qkine has optimized the SCF manufacture process to produce a highly bioactive protein with excellent lot-to-lot consistency for enhanced experimental reproducibility.
Human TPO – Qk098 - 25 µg
Thrombopoietin (TPO) is a cytokine which stimulates the production and differentiation of megakaryocytes, regulating platelet production.
Human VEGF – Qk048 - 25 µg
VEGF 165 is commonly used with human-induced pluripotent stem cells or embryonic stem cells-derived endothelial cells for developing human vascular tissue models.
Astrocyte differentiation kit (Qk520)

Human noggin - Qk034 - 50 µg
Noggin is used in the culture of intestinal, pancreatic, lung and tumor-derived organoids and the maintenance of undifferentiated embryonic stem cells (ESC) and for stem cell differentiation into neural and microglial lineages.
FGF2-G3 145 aa - Qk052 - 50 µg
A thermostable engineered form of FGF-2. FGF2-G3 145 aa is the 145 aa active domain of FGF-2 with the functional half-life increased from <10 h (wild-type) to >7 days (FGF2-G3).
Human EGF – Qk011 - 100 µg
Potent EGF-family growth factor used in many human and mouse organoid and stem cell culture systems including intestinal and tumor organoid culture.
Human CNTF - Qk063 - 25 µg
Used to culture primary neurons and glial cells such as astrocytes and Schwann cells. It is also used to culture retinal cells and adipocytes.
What's new?
Recombinant human IL-2 protein (Qk089-CTG)
Interleukin-2 (IL-2) is a vital cytokine that regulates immune responses by promoting the growth and activation of T-cells, enhancing immune function, and maintaining immune tolerance for immune activation and suppression. In cell therapy development IL-2 is used in the expansion of CAR-T cells and other T cell therapies.
Qkine has optimized the IL-2 manufacture process to produce a highly bioactive protein with excellent lot-to-lot consistency for enhanced experimental reproducibility. Cell therapy grade IL-2 is a highly pure animal origin-free 15.5 kDa monomer.
Qkine cell therapy grade high purity animal origin-free proteins are manufactured as GMP grade equivalents in an ISO 9001:2015-certified facility, under ISO 20399:2022 standards with GMP compliance, defined quality criteria and documentation.
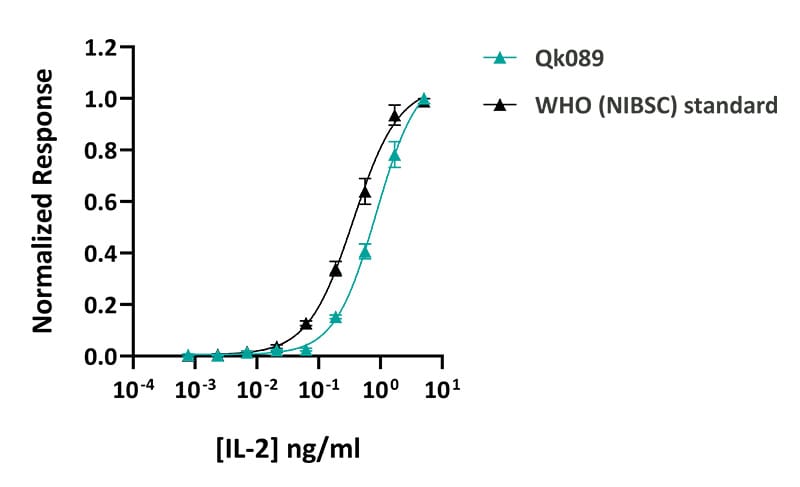
Qkine IL-2 was slightly less active compared to the WHO (NIBSC) standard IL-2. IL-2 activity was determined using a SRE luciferase reporter assay in transiently transfected HEK293 cells co-transfected with the IL-2 receptors (IL-2Rβ and IL-2Rγ). Transfected cells were treated in triplicate with a serial dilution of IL-2 for 3 hours. Firefly activity was measured and normalized to the control Renilla luciferase activity. Qk089 #204686 EC50 = 0.86 ng/ml, WHO (NIBSC) EC50 = 0.37 ng/ml.
Technotes
At Qkine data transparency is our promise. We test all new products against others on the market to ensure each is as bioactive or more, only then will they be approved for sale.
Each new lot is tested to ensure the bioactivity is consistent lot-to-lot, to ensure the most consistent cell cultures and assays. All data is available for all lots, and many are available as technotes.
Why not see if data on your protein of interest is available (if not ask!)
Technote | GM-CSF (Qk076)
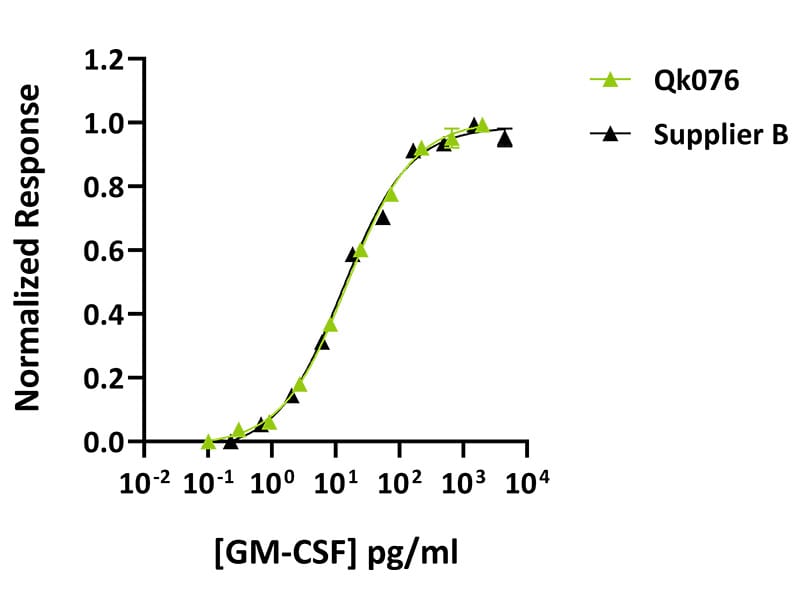
Qkine GM-CSF was found to be as bioactive as GM-CSF from an alternative major supplier. Bioactivity was determined using proliferation of TF-1 cells. Cells were treated in triplicate with a serial dilution of Qkine GM-CSF (Qk076, green) or Peprotech (300-03, Supplier B, black) for 48 hours. Protein concentrations were measured before use (Peprotech recovery 116%, Qkine 104%). Firefly luciferase activity is measured and normalized to control Renilla luciferase activity.
Technote | IL-7 (Qk095)
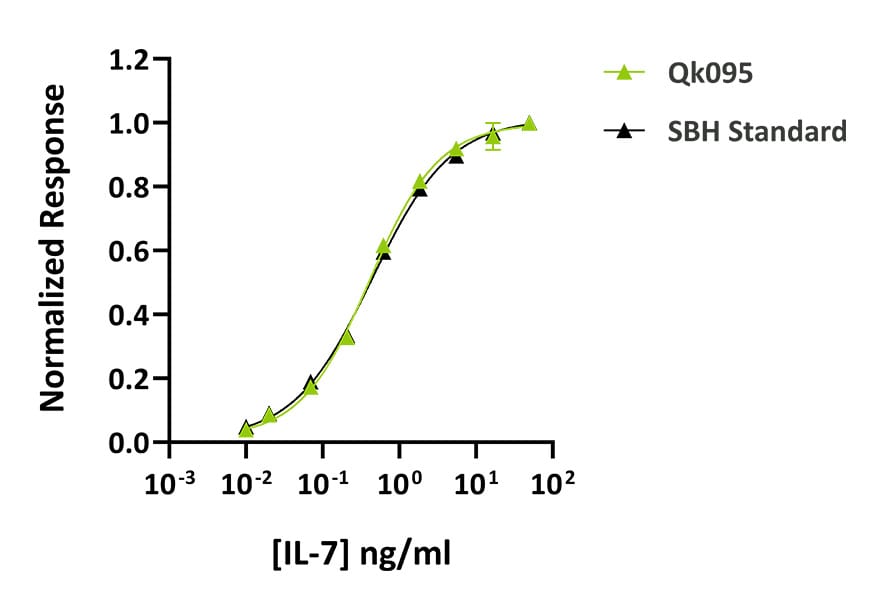
Qkine IL-7 was as bioactive as IL-7 from an alternative supplier. Recombinant IL-7 activity was determined using proliferation of the mouse-derived B lymphocyte cell line 2E8. Cells were treated in duplicate with a serial dilution of Qkine IL-7 (Qk095, green) for 65 hours. Cell viability was measured using the CellTiter 96 Aqueous Non-Radioactive Cell Proliferation Assay (Promega). Assay was performed by SBH Sciences using their standard (black) for comparison. Data from Qk095 lot #204669.
Recent publications
Signaling reprogramming via Stat3 activation unravels high-fidelity human post-implantation embryo modeling
Chen, C et al. Cell Stem Cell, October 2025
Human embryo models hold great promise for advancing medicine, but current systems lack efficiency and fidelity in replicating post-implantation stages. Here, we investigate whether STAT3 activation can reprogram pluripotent stem cells (PSCs) into early fates that self-organize into embryo models.
Tumor suppressor protein p53 governs human trophoblast lineage development
All our growth factors are manufactured within a stringent quality framework, ensuring high-quality proteins that maintain robust, reproducible, and physiologically relevant stem cell and organoid cultures that adhere to our Nine-point Quality Commitment.
Upcoming Events
-
- Mercia Stem Cell Alliance | 3 December 2025 | Keele, UK
- WORD+25 Research Celebration | 4 December 2025 | London, UK
- Assembloids & Complex Cell-Cell Interactions across Tissues & Organs | 8-11 December 2025 | CHSL, New York, USA
- ISSCR Boston International Symposium | 11-12 December 2025 | Cambridge, MA, USA
We'd always love to meet you at any of events, contact ryan.weber@matriqx.com to make an appointment.
What’s next?
Qkine are committed to producing the most high quality and bioactive growth factors and cytokines, coming soon to our extensive portfolio:
-
Recombinant human activin B protein (Qk024) - Expected January 2026
-
Recombinant human activin C protein (Qk026) - Expected January 2026
If you wish to be notified when proteins are released, contact us ryan.weber@matriqx.com