Differentiation of induced pluripotent stem cells (iPSCs) into astrocytes
Leighton Sneade
Qkine
Introduction
Astrocytes are the most abundant glial cells in the human central nervous system and are essential for maintaining healthy brain function. They regulate neurotransmitter uptake, ion and metabolic homeostasis, synaptic activity, and blood–brain barrier integrity, while also contributing to neuroimmune signaling, inflammation, injury response, and neuronal survival. Their ability to protect neurons from excitotoxicity, modulate synaptic networks, and maintain the extracellular environment makes them fundamental to overall CNS physiology [1].
Increasing evidence links astrocytic dysfunction to the pathogenesis of numerous neurological and neurodegenerative diseases, including Alzheimer’s disease, amyotrophic lateral sclerosis (ALS), Parkinson’s disease, and epilepsy. Consequently, physiologically relevant human astrocyte models are essential for advancing our understanding of CNS biology, disease mechanisms, and for drug discovery and neurotoxicity testing [2].
Given their extensive regulatory roles, astrocytes have become central to research on neurodevelopmental, neurodegenerative, and psychiatric disorders, as well as brain cancers. Dysfunction in astrocyte signaling, maturation, or metabolic support is now recognized as a contributing factor in conditions ranging from common neurodegenerative diseases to gliomas, underscoring the need for physiologically relevant human astrocyte models [3,4].
Human induced pluripotent stem cells (iPSCs) provide an ideal platform for generating such models. iPSC-derived astrocytes are genetically defined, renewable, and patient-specific, enabling controlled investigation of disease mechanisms, cell–cell interactions, developmental processes, and therapeutic responses. Their differentiation typically recapitulates in vivo developmental stages, including neural induction, radial glial specification, and gliogenesis, ultimately producing astrocytes expressing key markers such as GFAP, EAAT1/2, and ALDH1L1 [5].
This application note presents a streamlined and reproducible workflow for the generation of functional human astrocytes from iPSCs. The approach employs defined culture conditions and optimized media formulations to enhance efficiency, reduce differentiation time, and improve consistency. The resulting astrocytes exhibit characteristic morphology, marker expression, and functional properties, making them suitable for downstream applications in disease modeling, neuroinflammation studies, and high-content compound screening.
Methods
iPSC Maintenance
iPSCs were passaged twice per week using 0.5 mM EDTA for cell detachment and seeded onto vitronectin (Qk120)-coated (5 µg/ml) 6-well plates using a 1:6 split ratio and cultured in E8-like medium. The day after passage, spent media was removed and replaced with 5 ml of E8-like medium, enabling a weekend-free media change schedule. For further information on this process, please see our guide to weekend-free human induced pluripotent stem cell culture using thermostable FGF-2 (bFGF) from Qkine, together with our animal origin-free TGF-β1 and vitronectin, for improved colony homogeneity.
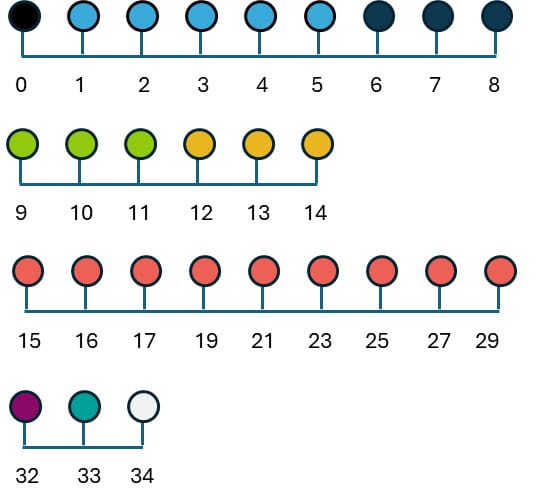
Figure 1. Schedule for astrocyte differentiation
Embryoid Body (EB) Formation (Day 0)
Human induced pluripotent stem cells (iPSCs) were maintained under standard conditions and used at approximately 70–80% confluency for differentiation (figure 1).
iPSCs were plated at a density of 5 × 10⁵ cells/ml in neural EB forming medium supplemented with 10 µM Y-27632 (ROCK inhibitor) (Table 1) to enhance cell survival. Cells were transferred to low-attachment plates and centrifuged at 800 ×g for 3 minutes to promote aggregation and formation of embryoid bodies (EBs).
| Component | Final Concentration |
| DMEM/F12 | 1x |
| N2 supplement | 1x |
| B27 supplement | 1x |
| GlutaMAX | 1x |
| Noggin (Qk034) | 500 ng/ml |
| FGF2-G3 (Qk052) | 4 ng/ml |
| SB431542 | 20 µM |
| Y-27632 (ROCK inhibitor) | 10 µM |
Table 1. Neural EB forming medium composition + ROCK inhibitor
Neural Induction (Days 1–5)
EBs were cultured in the same Neural EB forming medium formulation (Table 1), but without ROCK inhibitor. Media changes were performed carefully each day to avoid disrupting the aggregates. This stage promoted early neural induction and patterning.
Radial Glial Specification (Day 6)
EBs were dissociated into single cells using AccutaseTM and replated at 5 × 10⁵ cells/ml onto 0.5 µg/ml Matrigel-coated 6-well plates. Cells were cultured in Neurobasal-based medium 1 (Table 2) to promote radial glial precursor formation.
| Component | Final Concentration |
| Neurobasal medium (Gibco) | 1x |
| N2 supplement | 1x |
| FGF2-G3 (Qk052) | 10 ng/ml |
| EGF (Qk011) | 10 ng/ml |
Table 2. Neurobasal-based medium composition 1
Early Astrocyte Commitment (Days 7–11)
From Day 7 to Day 8, cells received daily media changes using the same Neurobasal-based medium 1 (Table 2).
From Day 9 to Day 11, CNTF (Qk063, 20 ng/ml) was also added to the Neurobasal-based medium 1 to initiate astrocyte lineage specification (Table 3).
| Component | Final Concentration |
| Neurobasal medium (Gibco) | 1x |
| N2 supplement | 1x |
| FGF2-G3 (Qk052) | 10 ng/ml |
| EGF (Qk011) | 10 ng/ml |
| CNTF (Qk063) | 20 ng/ml |
Table 3. Neurobasal-based medium composition 2
Astrocyte Differentiation (Days 12–29)
From Day 12 to Day 14, cells were maintained in Neurobasal-based medium 3, with daily media replacement (Table 4).
| Component | Final Concentration |
| Neurobasal medium (Gibco) | 1x |
| N2 supplement | 1x |
| FGF2-G3 (Qk052) | 10 ng/ml |
| CNTF (Qk063) | 20 ng/ml |
Table 4. Neurobasal-based medium composition 3
From Day 15 onward (Days 15, 16, 17, 19, 21, 23, 25, 27, and 29), media changes continued using the same formulation but without FGF2-G3 to promote astrocyte maturation (Table 5).
| Component | Final Concentration |
| Neurobasal medium (Gibco) | 1x |
| N2 supplement | 1x |
| CNTF (Qk063) | 20 ng/ml |
Table 5. Neurobasal-based medium composition 4
Immunocytochemistry and Imaging (Days 32–34)
On Day 32, cells were fixed and permeabilized prior to incubation with primary antibodies targeting astrocytic markers (i.e., GFAP, EAAT1, EAAT2, and ALDH1L1). On Day 33, secondary antibody staining was performed. From Days 34, cells were imaged using fluorescence microscopy to assess astrocyte morphology and marker expression.
Results
Immunocytochemistry (ICC) analysis demonstrated that the iPSC-derived cells acquired an astrocytic phenotype. After differentiation, cultures contained cells with large, star-shaped cell bodies and multiple elongated processes, consistent with the characteristic morphology of astrocytes in vitro. Marker staining confirmed expression of astrocyte-specific proteins (Figure 2): GFAP labeling was detected throughout the cytoplasm and processes, while EAAT1 and EAAT2 were localized to the plasma membrane. ALDH1L1 expression was also observed throughout the cytoplasm, providing further evidence of astrocytic identity.
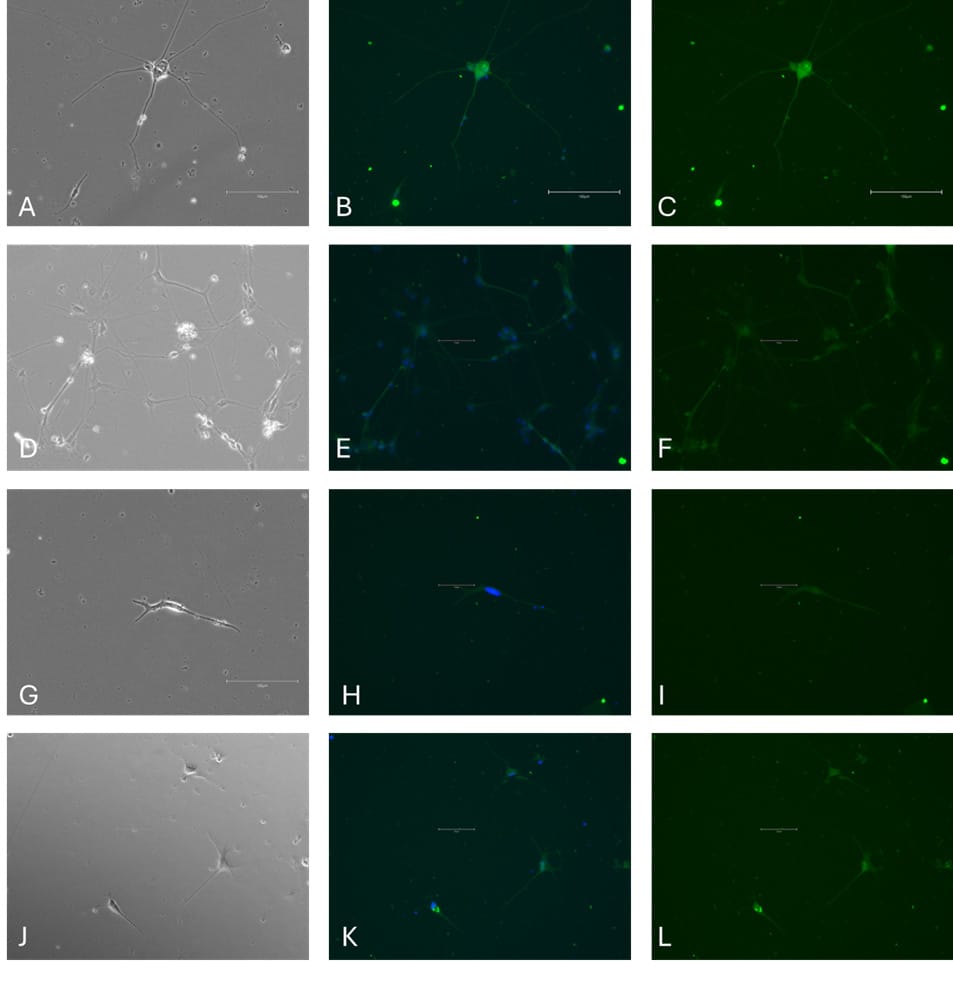
Figure 2. ICC of astrocyte markers in differentiated iPSCs. Phase contrast image (A); GFAP & Hoechst 33258 (green expression & blue expression, B); GFAP (green, C); Phase contrast image (D); ALD1L1 & Hoechst 33258 (green expression & blue expression, E); ALD1L1 (Green, F); Phase contrast image (G); EAAT1 & Hoechst 33258 (green expression & blue expression H); EAAT1 (green, I). Phase contrast image (J); EAAT2 & Hoechst 33258 (green expression & blue expression, K); EAAT2 (green, L). Scale bar = 150 µm.
Conclusion
The growing significance of iPSCs in clinical and research applications stems from their unique ability to differentiate into all somatic cell types in the human body. Successful differentiation into specific lineages, such as astrocytes, depends on maintaining pluripotency during culture and achieving homogenous, reproducible differentiation. The use of optimized medium containing highly pure and bioactive growth factors is critical to driving efficient, high-quality differentiations and outcomes.
The data presented in this application note demonstrate that high purity, animal origin-free growth factors from Qkine – including EGF (Qk011), Noggin (Qk034), FGF2-G3 (Qk052) and CNTF (Qk063) – support robust and reliable differentiation of iPSCs into astrocyte cells. These growth factors reduce variability and enhance reproducibility, which are essential for both research and therapeutic applications.
Astrocyte cells were confirmed by immunocytochemistry, showing expression of key markers. The use of a weekend-free culture system prior to differentiation, enabled by thermostable FGF-2 (Qk052) and used with animal origin-free vitronectin (Qk120) and TGF-β1 (Qk010), further contributed to consistent colony morphology and reduced handling variability. For further information on this process, please see our guide to Weekend-free human induced pluripotent stem cell culture. This work highlights the importance of high-quality reagents in overcoming challenges associated with iPSC differentiation, such as inconsistent cell performance or variability in outcome. By using Qkine’s portfolio of defined, animal origin-free growth factors from Qkine, researchers can improve the reproducibility of complex, long-term cell cultures and advance applications in regenerative medicine, drug discovery, and disease modeling.
Further information
Qkine growth factors are manufactured to the highest quality standards and are free from animal-derived contaminants, delivering low endotoxicity and high purity. At Qkine, we are committed to raising the standards of growth factors, cytokines and related proteins to better support long-term and complex cell cultures. We are a science-led team, please reach out with any questions or requests to ryan.weber@matriqx.com.
References
[1] Emdad L, D’Souza SL, Kothari HP, Qadeer ZA, Germano IM. Efficient differentiation of human embryonic and induced pluripotent stem cells into functional astrocytes. Stem Cells Dev. 2012 Feb 10;21(3):404-10. doi: 10.1089/scd.2010.0560.
[2] Stoklund Dittlau, K.; Freude, K. Astrocytes: The Stars in Neurodegeneration? Biomolecules 2024, 14, 289. https://doi.org/10.3390/biom14030289
[3] Jovanovic VM, Weber C, Slamecka J et al. A defined roadmap of radial glia and astrocyte differentiation from human pluripotent stem cells. Stem Cell Reports. 2023 Aug 8;18(8):1701-1720. doi: 10.1016/j.stemcr.2023.06.007.
[4] Tcw J, Wang M, Pimenova AA, Bowles KR et al. An Efficient Platform for Astrocyte Differentiation from Human Induced Pluripotent Stem Cells. Stem Cell Reports. 2017 Aug 8;9(2):600-614. doi: 10.1016/j.stemcr.2017.06.018.
[5] Voulgaris, D., Nikolakopoulou, P. & Herland, A. Generation of Human iPSC-Derived Astrocytes with a mature star-shaped phenotype for CNS modeling. Stem Cell Rev and Rep 18, 2494–2512 (2022). https://doi.org/10.1007/s12015-022-10376-2
All our recombinant proteins are animal-free and come with Bioactivity. Guaranteed.
Featured resources
Contact us
Our science team is here to help, please contact us if you have any questions.