Commercial versus in-house media: A comparative study of human induced pluripotent stem cell maintenance
Leighton Sneade
Qkine
Introduction
Since their initial description, human iPSCs have transformed biomedical research by enabling patient-specific disease modeling and providing a renewable source of cells for drug screening and regenerative therapies [1]. The maintenance of iPSCs in an undifferentiated and genetically stable state demands culture conditions that accurately replicate essential signaling environments while minimizing the use of undefined components. Early feeder-dependent systems relying on mouse embryonic fibroblasts have largely been replaced by feeder-free, chemically defined media, reducing variability and enhancing clinical applicability.
Commercial Media Formulations
Several commercial media have achieved widespread adoption:
- TeSR™-E8™ / Essential 8 (E8): Minimal formulations comprising only eight defined components were developed to sustain iPSCs without serum or feeder layers. These media maintain the expression of pluripotency marker, stable karyotypes, and differentiation potential over extended passages [1].
- NutriStem™: NutriStem XF (Sartorius) is a fully defined, xeno-free system designed to minimize protein content while preserving the capacity for self-renewal [2].
These media are produced under current Good Manufacturing Practice (cGMP) conditions and validated across numerous lines, providing reproducibility for both academic and clinical research settings.
In-House Formulations
Academic groups have developed open-source media as cost-efficient alternatives.
- B8 Medium: This chemically defined medium substitutes costly components with optimized concentrations of insulin, transferrin, and ascorbic acid, and includes a thermostable FGF2 variant and TGF-β3. B8 supports long-term culture of human iPSCs with preserving karyotypic integrity and the expression of pluripotency markers [3].
- Refined Variants (E8-like medium): An in-house Qkine developed medium, similar to E8, that has demonstrated a comparable capacity to maintain pluripotency. This was validated in the application note describing Weekend-free human induced pluripotent stem cell culture.
While in-house media significantly reduce costs, they require rigorous quality control and may exhibit variability in scalability, consistency, and robustness.
This study aims to directly compare the performance of selected commercially available iPSC media with in-house formulations. Key parameters assessed include cell morphology and proliferation rate. By systematically evaluating both types of media, we aim to provide practical insights for laboratories seeking to balance cost, performance, and experimental reproducibility in human iPSC culture systems.
| Media Type | Cost per 500 ml |
| Essential 8 (E8) | £268.00 |
| TeSR E8 | £248.00 |
| NutriStem® hPSC XF GF-free + Qkine FGF2-G3 and TGF-β1 | £259.20 |
| E8-like media + Qkine FGF2-G3 and TGF-β1 | £110.12 |
Table 1. Cost per 500 ml for commonly used iPSC media, correct as of August 2025.
Methods
Cell Culture and Maintenance
Human iPSCs were maintained under feeder-free conditions and passaged twice weekly using 0.5 mM EDTA for detachment. Cells were seeded at a 1:6 split ratio onto vitronectin–coated 6-well plates (Qk120; 5 µg/ml) and cultured in an E8-like medium. To enable a weekend-free culture schedule, spent medium was replaced with 5 ml of fresh E8-like medium on the day following passage. For further details, refer to our guide on Weekend-free human induced pluripotent stem cell culture using thermostable FGF-2 (bFGF) (Qk052 or Qk053) in combination with animal origin-free TGF-β1 (Qk010) and vitronectin (Qk120), which supports improved colony homogeneity.
Media Adaptation
Following plating, iPSCs were transitioned to the test media formulations. Cells were cultured for two weeks under these conditions to allow adaptation prior to comparative evaluation. NutriStem™ had Qkine TGF-β1 (Qk010) and FGF2-G3 (145aa) (Qk052) added to complete the medium. E8 and TeSR™-E8™ media were media changed daily throughout the testing period.
Colony Growth Assay
Adapted iPSCs were passaged with 0.5 mM EDTA and reseeded at a 1:6 split ratio on vitronectin–coated 6-well plates (Qk120; 5 µg/ml) in their respective test media. After a 15-minute settling period, plates were placed on the Axion Omni live-cell imaging system and imaged hourly. The following day, the spent medium was replaced with 5 ml of fresh test medium for NutriStem™ and E8-like media variants, which were then imaged continuously throughout the experimental period, or 2 ml of fresh E8 and TeSR™-E8™ daily, ensuring the imaging process was not disrupted, after which hourly imaging continued.
Single-Cell Plating Assay
Adapted iPSCs were dissociated using Accutase™ and seeded at 1,000 cells per well onto vitronectin-coated 24-well plates (Qk120; 5 µg/ml). Cells were cultured in their respective test media supplemented with 10 µM ROCK inhibitor (Y-27632). Following a 15-minute settling period, plates were transferred to the Axion Omni and imaged hourly. On the following day, cells were fed with fresh medium lacking ROCK inhibitor, timed to avoid disruption of scheduled imaging sessions, as described in the Colony Growth Assay section.
Results
In-house E8-like culture medium formulation incorporating Qkine FGF2-G3 (145 aa) and TGF-β1 supported the maintenance of human iPSCs at levels comparable to the commercially available TeSR™-E8™ and E8 media. Typical undifferentiated colony morphology was observed with in-house, TeSR™-E8™ and E8 media (figures 1 and 2). iPSC proliferation rates were also similar on both 6 well (figure 3) and 24 well (figure 4) plates, qualified using the Axion Omi system. No significant differences were observed in growth kinetics or colony integrity between cells cultured in the E8-like medium and those maintained in TeSR™-E8™ or Essential 8.
Typical compact colony morphology was not initially observed in the NutriStem™ media (figures 1 and 2, C and F), where colonies initially expanded into spiky morphology before compacting and reverting to the more common compact morphology and rounded colonies. These larger cells forming the spikey colonies in NutriStem™ looked like an initially higher proliferation rate in this media on the quantification (figures 3 and 4), but this levelled off as the colonies rounded into the typical iPSC morphology. These results indicate that the Qkine FGF2-G3/TGF-β1 formulation provides an effective, defined, and xeno-free system for iPSC culture.

Figure 1. Colony morphology 48 hours after colony passage.
Representative images of iPSCs cultured in: (A) E8-like medium with FGF2-G3 (145 aa) (Qk052); (B) E8-like medium with FGF2-G3 (154 aa) (Qk053); (C) NutriStem™ with FGF2-G3 (145 aa) (Qk052); (D) Life Technologies E8; (E) TeSR-E8; (F) NutriStem™ with FGF2-G3 (154 aa) (Qk053). Images were acquired using the Axion Omni live-cell imaging system. Typical compact colony morphology was observed under all conditions except NutriStem™, where colonies initially expanded into spiky morphologies before reverting to smaller, tighter, rounded colonies.
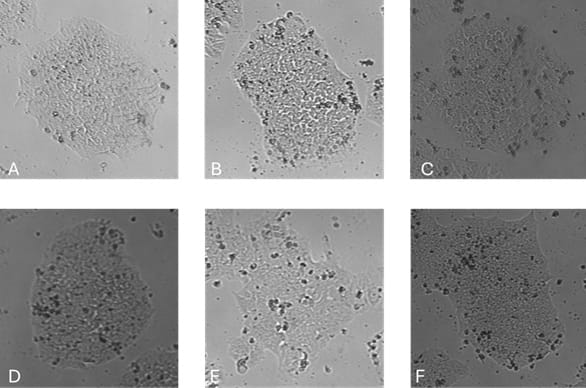
Figure 2. Colony morphology 48 hours after Single cell passage.
Representative images of iPSCs cultured in: (A) E8-like medium with FGF2-G3 (145 aa) (Qk052); (B) E8-like medium with FGF2-G3 (154 aa) (Qk053); (C) NutriStem™ with FGF2-G3 (145 aa) (Qk052); (D) Life Technologies E8; (E) TeSR-E8; (F) NutriStem™ with FGF2-G3 (154 aa) (Qk053). Images were acquired using the Axion Omni live-cell imaging system. Typical compact colony morphology was observed under all conditions except NutriStem™, where colonies initially expanded into spiky morphologies before reverting to smaller, tighter, rounded colonies.
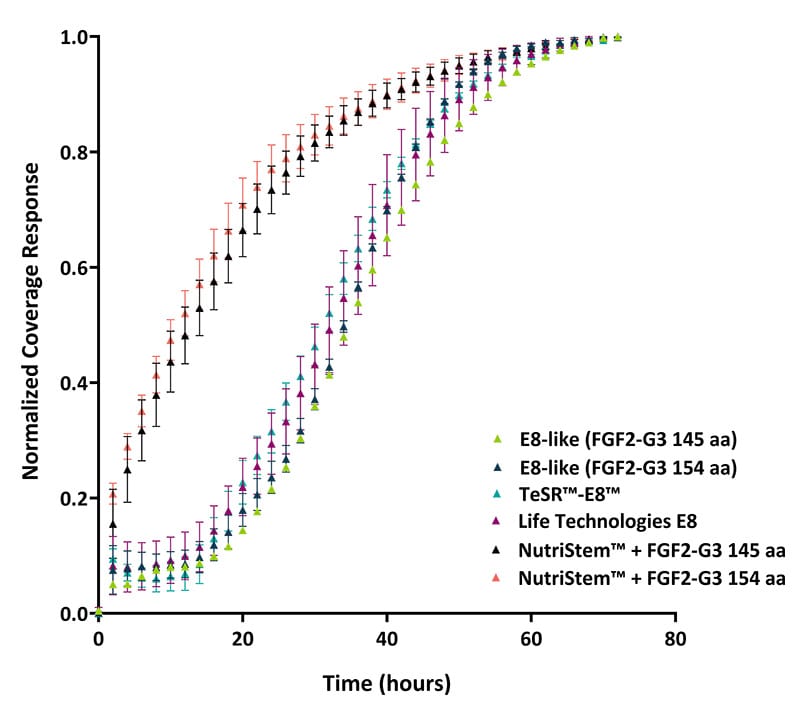
Figure 3. Growth rate analysis based on well coverage in a 6-well plate (measured in triplicate). Growth rates were assessed using the Axion Omni live-cell imaging system. An increased growth rate was observed with NutriStem™ medium, attributed to colonies expanding outward to occupy the available well space prior to forming tightly packed colonies.
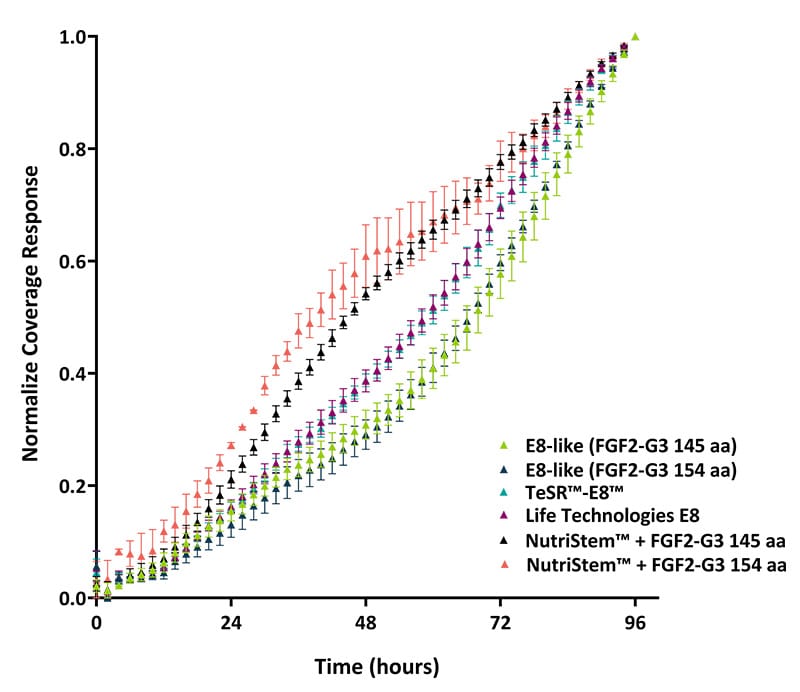
Figure 4. Growth rate analysis based on well coverage in a 24-well plate (measured in triplicate). Growth rates were assessed using the Axion Omni live-cell imaging system. An increased growth rate was observed with NutriStem™ medium, attributed to colonies expanding outward to occupy the available well space prior to forming tightly packed colonies.
Conclusion
In this study, we evaluated multiple culture media for the maintenance of human induced pluripotent stem cells (iPSCs). Our results demonstrate that an E8-like formulation incorporating Qkine TGF-β1 (Qk010) and thermostable FGF-2 (bFGF) (Qk052 or Qk053) supports iPSC maintenance at a level comparable to the xeno-free, chemically defined TeSR™-E8™ and Essential 8 (E8) media. When used in combination with recombinant human vitronectin (Qk120), this medium preserved iPSC proliferation rates and typical colony morphology.
The defined conditions presented here enhance experimental reproducibility and consistency in iPSC culture, both in basic research and in translational contexts. Furthermore, the capacity to sustain long-term iPSC culture without compromising safety or functionality provides a cost-effective strategy for large-scale expansion, thereby facilitating a wide range of downstream applications.
References
- Chen G, Gulbranson DR, Hou Z, et al. Chemically defined conditions for human iPSC culture: the Essential 8 medium. Nat Methods. 2011;8(5):424-429. doi: 10.1038/nmeth.1593
- Bergström, R., Ström, S., Holm, F., Feki, A., Hovatta, O. (2011). Xeno-Free Culture of Human Pluripotent Stem Cells. In: Schwartz, P., Wesselschmidt, R. (eds) Human Pluripotent Stem Cells. Methods in Molecular Biology, vol 767. Humana Press. https://doi.org/10.1007/978-1-61779-201-4_9
- Lyra-Leite DM, et al. B8 medium: a chemically defined, cost-efficient formulation for human iPSC maintenance. Stem Cell Reports. 2020;14:1-12. doi: 10.1002/cpsc.110
Further Information
These iPSC have then been differentiated into various germ layer and cell types, further indicating the fact that their pluripotency is maintained using our weekend-free media change conditions in combination with our growth factors.
When compared to iPSC cultures grown in an E8-type media, those cells demonstrate a similar proliferation rate but with higher levels of spontaneous differentiation, lowering the quality of the iPSC colonies for future work. These cells also required daily media exchange, resulting in higher cost and time investment in maintaining the cultures (data available upon request).
Qkine growth factors are manufactured to the highest of quality standards and are free from animal-derived contaminants, delivering low endotoxicity and high purity. At Qkine, we are committed to raising the standards of growth factors, cytokines and related proteins to better support long-term and complex neural stem cell culture. We are a science-led team, please reach out with any questions or requests to ryan.weber@matriqx.com.
All our recombinant proteins are animal-free and come with Bioactivity. Guaranteed.
Featured resources
Contact us
Our science team is here to help, please contact us if you have any questions.