Enabling robust iPSC maintenance with QkPlate© pre-prepared dried vitronectin-coated plates
Leighton Sneade
Qkine
Induced pluripotent stem cells (iPSCs) represent a transformative platform in regenerative medicine, disease modeling, and drug discovery because of their capacity for unlimited self-renewal and multilineage differentiation [1]. A critical determinant of robust iPSC culture in vitro is the composition of the extracellular environment, particularly the substrate on which cells adhere and proliferate. Historically, feeder layers or undefined matrices such as Matrigel have supported iPSC adhesion; however, these systems entail significant batch variability and xenogeneic components that limit reproducibility and translational potential. A defined, xeno-free substrate is therefore essential for standardized cultured iPSC expansion and downstream applications.
Vitronectin, a glycoprotein abundant in the natural extracellular matrix and blood, has emerged as a key defined culture substrate that supports the adhesion, growth, and maintenance of pluripotency in human iPSCs under feeder-free, chemically defined conditions. Recombinant human vitronectin, when utilized as a coating in conjunction with defined media such as Essential 8 (E8), facilitates stable iPSC attachment via integrin-mediated interactions and sustains proliferation without feeder cells [2,3]. Importantly, vitronectin-coated cultures maintain normal karyotype and pluripotency marker expression across multiple passages, underscoring its utility for long-term culture and experimental consistency [4].
Mechanistically, vitronectin interacts with integrins including αvβ5 and β1 on the iPSC surface to promote adhesion and survival, enabling signaling contexts that support self-renewal [5]. In defined culture systems, this receptor engagement is fundamental to robust expansion, particularly when compared to undefined substrates where matrix composition can vary markedly. While alternative matrices such as laminin or collagen have been explored, vitronectin offers a balance of defined chemistry, reproducibility, and relative cost-effectiveness for routine iPSC culture and is compatible with scalable, clinical-grade workflows.
Given the increasing demand for reproducible and clinically relevant iPSC culture platforms, the adoption of vitronectin coating represents a critical advancement in establishing feeder-free, xeno-free culture environments that underpin reliable iPSC growth and downstream differentiation.
Here, we demonstrate the use and robustness of the QkPlate© human vitronectin‑coated six‑well plate (Qk2001) for the maintenance of iPSCs, in which pluripotency is preserved.
Methods
QkPlate© (Qk2001) preparation
Recombinant human vitronectin (Qk120) was coated at 5 µg per well in a 6-well tissue culture plate, and the plates dried under aseptic conditions. The plates were then vacuum-sealed and stored at room temperature until use.
Cell culture and maintenance
iPSCs were passaged twice weekly using 0.5 mM EDTA for detachment and seeded onto vitronectin coated 6-well plates using a 1:6 split ratio when cultures reached between 70-85% confluency. Cells were cultured in E8-like medium (Table 1). The day after passaging, the spent medium was removed and replaced with 5 ml of fresh E8-like medium. Cultures were maintained following a weekend-free schedule (Figure 1), full protocol.

Figure 1. Weekly schedule for routine maintenance of iPSC.
| Component | Final Concentration |
|---|---|
| DMEM/F12 medium (Thermo Fisher Scientific,31330038) | 1x |
| Sodium bicarbonate (Merck, S5761) | 543 mg/l |
| Sodium chloride (Merck, S5886) | 1000 mg/l |
| L-ascorbic acid (Merck, A8960) | 64 mg/l |
| Insulin-Transferrin-Selenium (ITS-G) (Thermo Fisher Scientific, 41400045) | 1x |
| FGF2-G3 (Qkine, Qk053) | 40 ng/ml |
| TGF-β1 PLUS (Qkine, Qk010) | 2 ng/ml |
Table 1. E8-like media construction components. Components added aseptically and filter sterilized before use.
Immunocytochemistry
iPSCs were passaged using Accutase for detachment and seeded at 500 cells/well in a vitronectin (5 µg/ml) coated 96-well plate in E8-like medium containing ROCK inhibitor (Y-27632, 10 µM). The day after seeding, the ROCK inhibitor was removed and 200 µl of E8-like medium was added to each well. After 3 days, cells were fixed with 4% Paraformaldehyde, blocked and permeabilized with 10% donkey serum diluted in 0.1% Triton X-100. Specific antibodies for markers of pluripotency (OCT-4, NANOG and SOX2) were then used to immunostain the cells overnight at 4 °C. iPSCs were then washed and incubated with secondary antibody Donkey anti-Mouse AlexaFluor 647 or Donkey anti-Goat AlexaFluor 488 and Hoechst 33258, followed by imaging in phosphate buffered saline (PBS). Brightfield and fluorescence images were acquired using the EVOS M5000 system.
Results
The attachment efficiency and subsequent proliferation of the iPSC line were not adversely affected when cells were cultured on human vitronectin pre-coated QkPlates (Qk2001). Cells exhibited robust adherence shortly after seeding and maintained sustained growth over multiple weeks in culture, with no observable differences in morphology or expansion kinetics compared to standard culture conditions (Figures 2-5). Throughout the culture period, the iPSC line retained characteristic features of pluripotent stem cells, including compact colony morphology with defined borders. Colonies cultured on dried plates were comparable to ones cultured on freshly prepared plates (Figures 7, 8). Consistent with this phenotype, the expression of established pluripotency-associated markers was preserved, indicating maintenance of the pluripotent state despite extended culture on dried coated substrates (Figure 6).
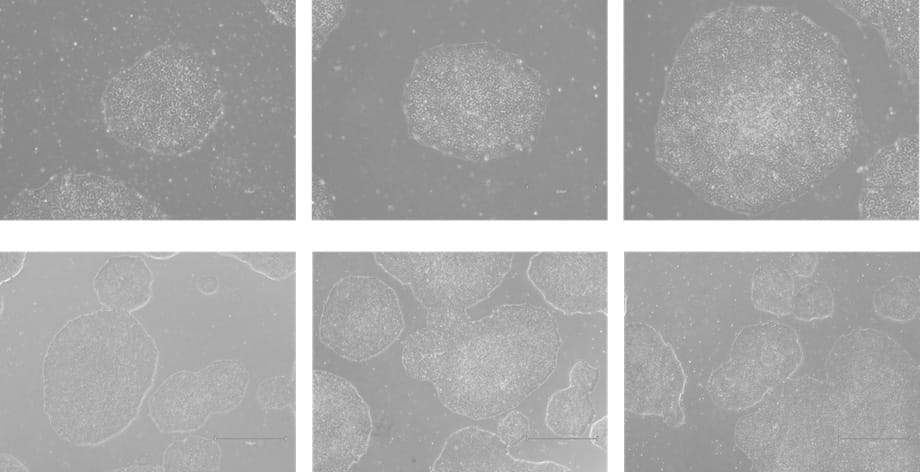
Figure 2. Phase-contrast images of WTSIi_005A cells cultured on human vitronectin pre-coated QkPlates (Qk2001) that had been dried and stored at room temperature for 2 weeks. Images are shown at two magnifications, with scale bars of 300 µm (top row) and 750 µm (bottom row).
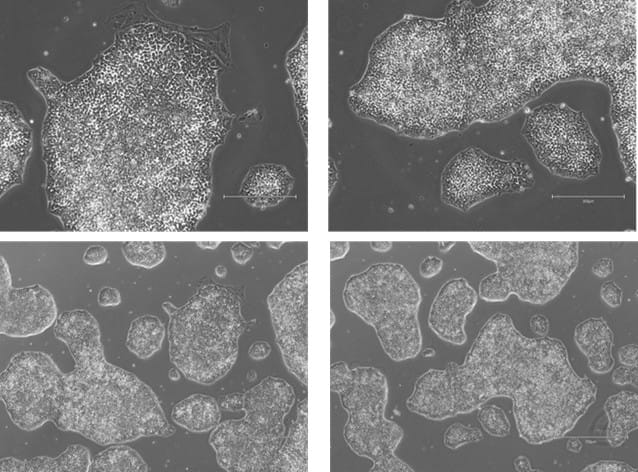
Figure 3. Phase-contrast images of WTSIi_005A cells cultured on human vitronectin pre-coated QkPlates (Qk2001) that had been dried and stored at room temperature for 1 month. Images are shown at two magnifications, with scale bars of 300 µm (top row) and 750 µm (bottom row).
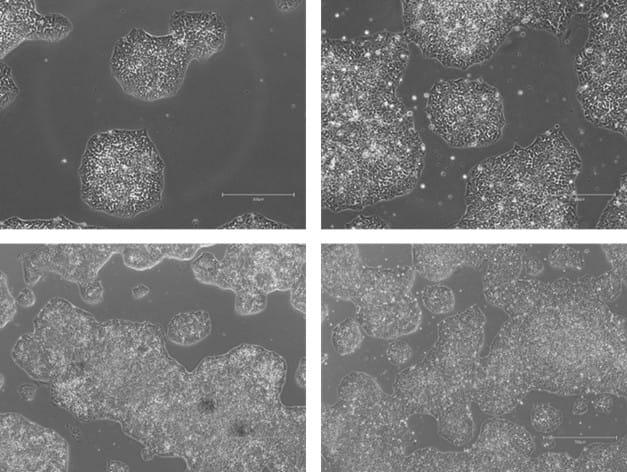
Figure 4. Phase-contrast images of WTSIi_005A cells cultured on human vitronectin pre-coated QkPlates (Qk2001) that had been dried and stored at room temperature for 3 months. Images are shown at two magnifications, with scale bars of 300 µm (top row) and 750 µm (bottom row).
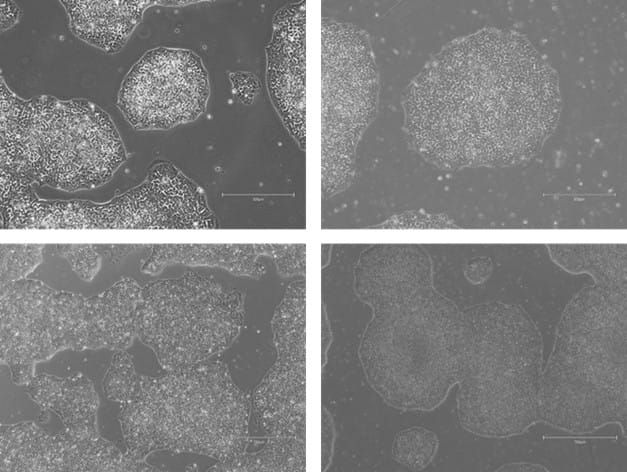
Figure 5. Phase-contrast images of WTSIi_005A cells cultured on human vitronectin pre-coated QkPlates (Qk2001) that had been dried and stored at room temperature for 6 months. Images are shown at two magnifications, with scale bars of 300 µm (top row) and 750 µm (bottom row).
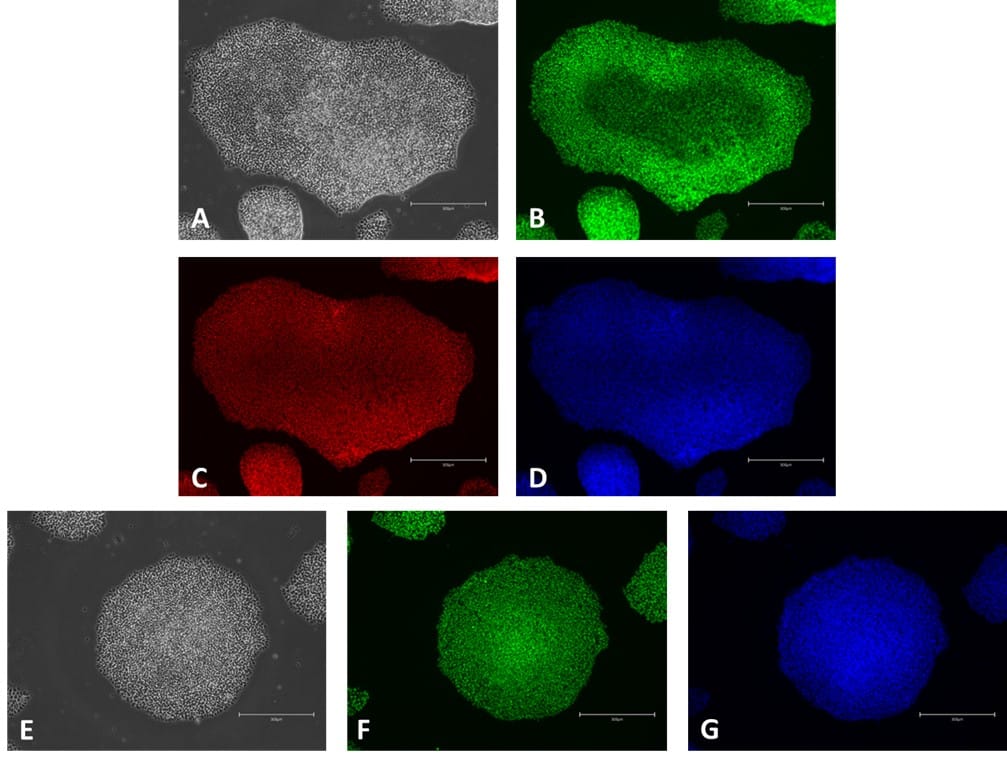
Figure 6. ICC of pluripotency markers in iPSC grown in E8-like medium on Qkplates (Qk2001). Phase contrast image (A); NANOG expression (green, B); OCT-4 expression (red, C); Hoechst 33258 (blue, D); Phase contrast (E); SOX2 expression (green, F); Hoechst 33258 (blue, G). Scale bar = 300 µm.
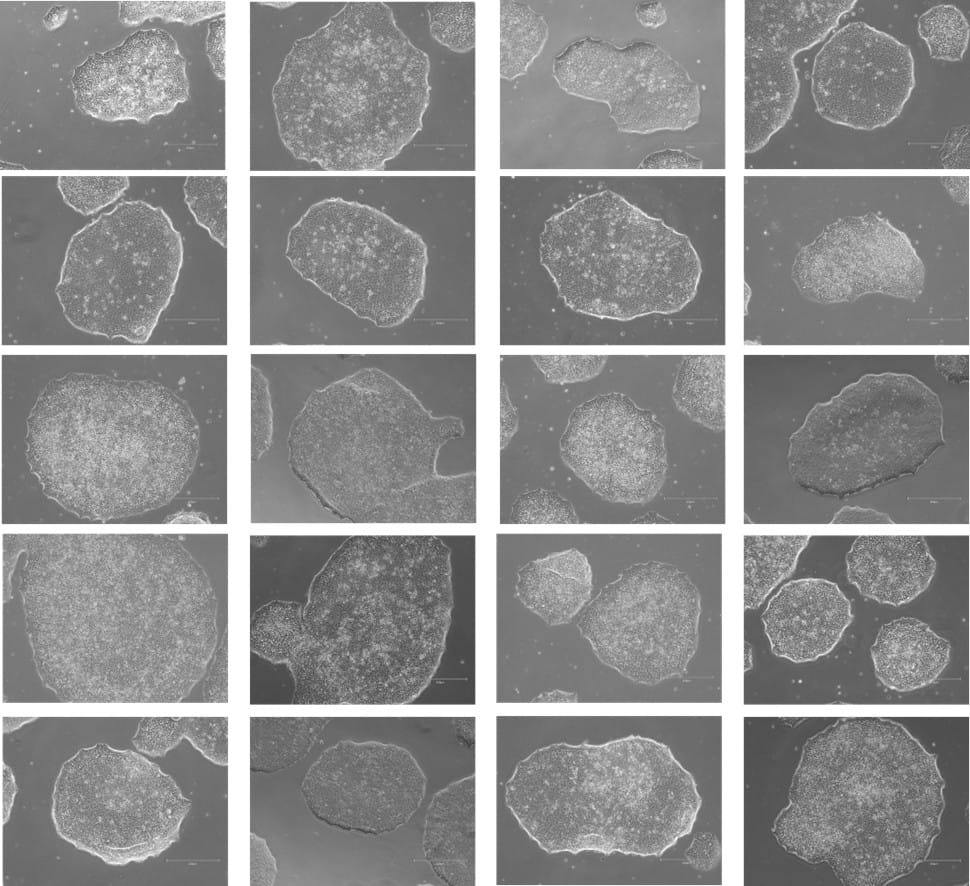
Figure 7. Phase-contrast images of WTSIi_005A cells acquired over a 10-week period of routine maintenance passaging. Cells were cultured on human vitronectin pre-coated QkPlates (Qk2001) that had been dried and stored at room temperature. Images illustrate sustained cell attachment, typical morphology, and stable growth over extended culture.
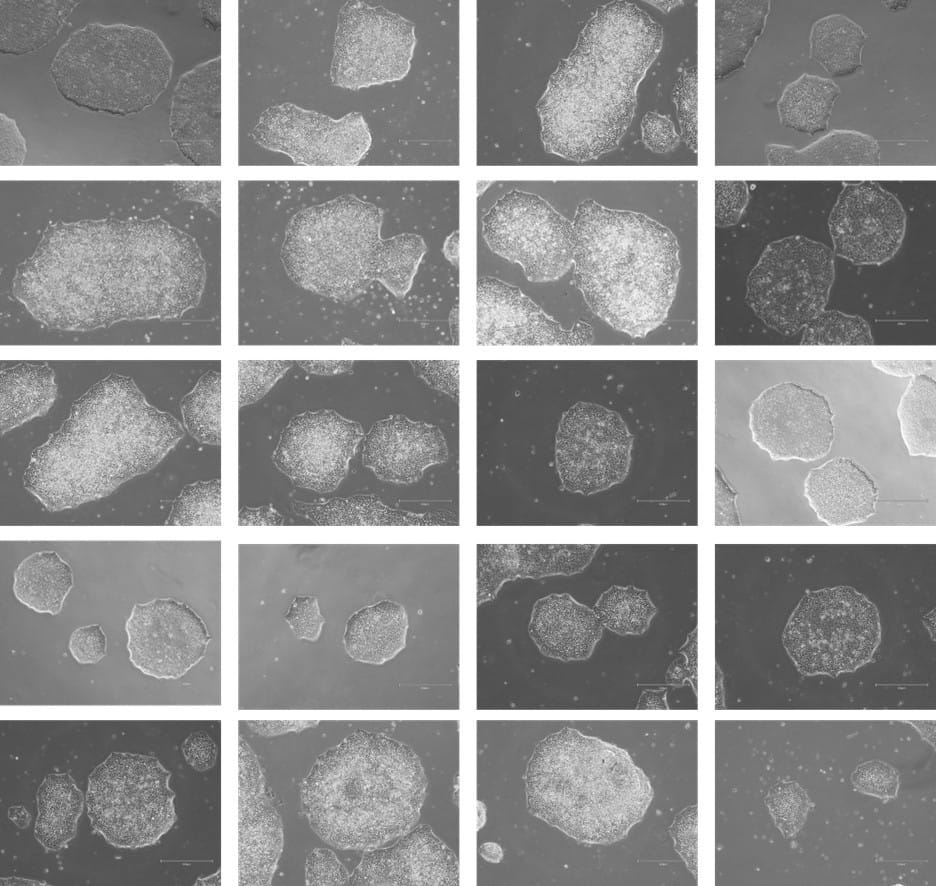
Figure 8. Phase-contrast images of WTSIi_005A cells acquired over a 10-week period of routine maintenance passaging. Cells were cultured on human vitronectin pre-coated QkPlates (Qk2001) that had been freshly prepared on the day of use. Images illustrate sustained cell attachment, typical morphology, and stable growth over extended culture.
Conclusion
The importance of iPSC in clinical and research fields is increasing due to their potential to differentiate into all somatic cell types in the human body. Successful differentiation depends on maintaining the pluripotency of iPSC during culture, making the use of the correct media crucial.
The data presented in this application note demonstrates that using human vitronectin pre-coated QkPlates (Qk2001) can preserve iPSC pluripotency and support proliferation while preventing the need for routinely coating of 6-well plates. Qkine’s dried vitronectin-coated plates retained key functional characteristics over extended maintenance, including stable attachment, sustained proliferation, and preservation of pluripotency. Cells maintained on these coated substrates were subsequently differentiated into derivatives of all three germ layers, further demonstrating that the dried vitronectin coating supports long-term maintenance of pluripotent capacity in combination with the applied culture conditions.
When compared with iPSC cultures maintained on freshly coated plates under E8-like medium conditions, cells cultured on dried vitronectin-coated plates exhibited comparable proliferation rates while displaying similarly low levels of spontaneous differentiation, resulting in retained colony morphology and overall culture quality for downstream applications. In addition, the use of pre-prepared dried coated plates enabled more flexible handling and reduced operational burden, contributing to lower time and resource investment during routine culture.
References
[1] Takahashi K, Tanabe K, Ohnuki M et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell (2007). doi: 10.1016/j.cell.2007.11.019
[2] Chen G, Gulbranson DR, Hou Z et al. Chemically defined conditions for human iPSC derivation and culture. Nat Methods. (2011). doi: 10.1038/nmeth.1593
[3] Badenes SM, Fernandes TG, Cordeiro CS et al. Defined Essential 8™ Medium and Vitronectin Efficiently Support Scalable Xeno-Free Expansion of Human Induced Pluripotent Stem Cells in Stirred Microcarrier Culture Systems. PLoS One (2016). doi: 10.1371/journal.pone.0151264
[4] Kaini RR, Shen-Gunther J, Cleland JM et al. Recombinant Xeno-Free Vitronectin Supports Self-Renewal and Pluripotency in Protein-Induced Pluripotent Stem Cells. Tissue Eng Part C Methods (2016). doi: 10.1089/ten.TEC.2015.0180
[5] Braam SR, Zeinstra L, Litjens S et al. Recombinant vitronectin is a functionally defined substrate that supports human embryonic stem cell self-renewal via alphavbeta5 integrin. Stem Cells (2008). doi: 10.1634/stemcells.2008-0291
Further Information
Qkine growth factors are manufactured to the highest of quality standards and are free from animal-derived contaminants, delivering low endotoxicity and high purity. At Qkine, we are committed to raising the standards of growth factors, cytokines and related proteins to better support long-term and complex stem cell culture. We are a science-led team, please reach out with any questions or requests to ryan.weber@matriqx.com.
All our recombinant proteins are animal-free and come with Bioactivity. Guaranteed.
Contact us
Our science team is here to help, please contact us if you have any questions.