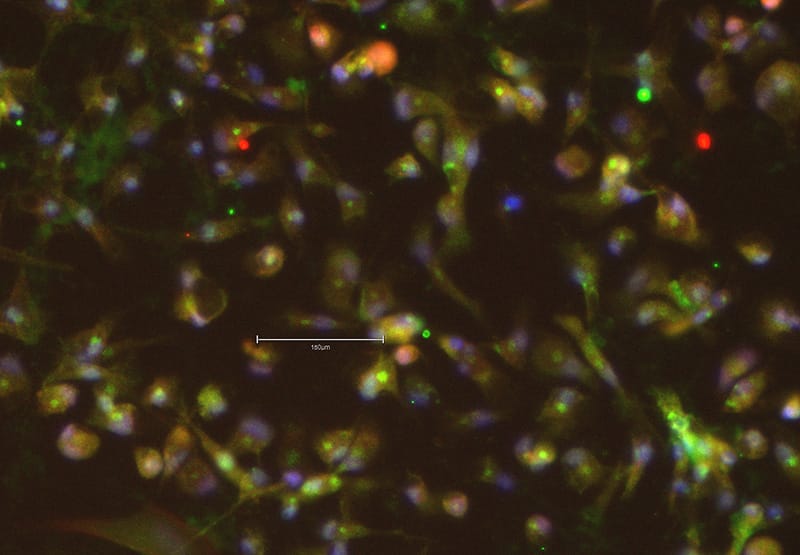
Commercially available media – the hidden pitfalls of proprietary formulations
Commercial media preparations can be a convenient option for stem cell and organoid maintenance and differentiation, but the proprietary 'black box' formulation prohibits trouble-shooting and optimization read our blog, as we ask, what's in it?