Pep talk - November 2025

Raising the standard: Developing a high-quality IL-34 protein
Interleukin-34 (IL-34) is a key growth factor involved in the development and regulation of monocytes and macrophages, cells that play essential roles in immunity, inflammation, and tissue repair. It’s also crucial for supporting microglia differentiation, making it especially important for researchers working in neuroscience, immunology, oncology, and drug development.
Despite its importance, scientists have long struggled to access a reliable, animal-free, and high-quality version of IL-34. Most available proteins on the market either don’t work well or vary too much from batch to batch. This makes experiments harder to reproduce and wastes both time and resources, something researchers simply can’t afford.
When we first started working on IL-34 at Qkine, we weren’t just trying to make another recombinant protein. We wanted to solve a real problem. We knew that if we could develop an IL-34 that was consistent, highly active, and free from animal-derived components, it could have a big impact on research.
Read more about our journey to a highly consistent and bioactivity IL-34
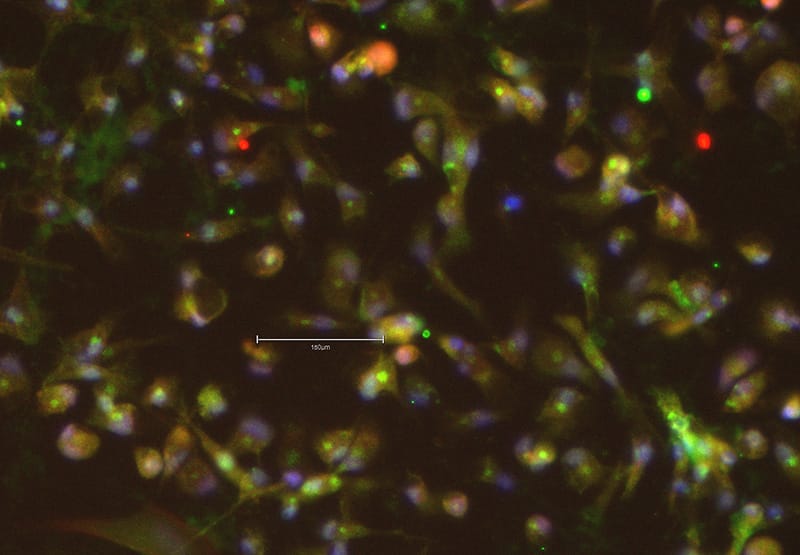
Staining of microglial markers after differentiation from iPSC with Qkine IL-34 PLUS™ (Qk091)

Application Note: Differentiation of induced pluripotent stem cells (iPSCs) into microglia
Microglia, which originate from mesodermal and mesenchymal lineage, serve as the primary innate immune cells of the central nervous system (CNS). They play crucial roles in synaptic plasticity, neurogenesis, maintaining homeostasis, and immune defense within the CNS. Studying human microglia has historically been challenging due to the difficulty of obtaining primary cells from human donors. To overcome this limitation, deriving microglia from induced pluripotent stem cells (iPSCs) has emerged as a promising solution.
In this application note, we demonstrate that high purity, animal origin-free growth factors from Qkine - including our new IL-34 PLUSTM (Qk091) along with BMP-4 (Qk038), FGF2-G3 (Qk052), SCF (Qk078), VEGF 165 (Qk048), IL-3 (Qk090), M-CSF (Qk075), Flt3 ligand (Qk087) and GM-CSF (Qk076) - support robust and reliable differentiation of iPSCs into microglial cells.
View and download the full application note
Qkine launches cell therapy grade recombinant proteins to support next-generation therapies
At the start of this October Qkine launched its new Cell Therapy Grade range of proteins with extended QC to be suitable for cell and gene therapy manufacturing.
Cell therapy is rapidly transforming modern medicine, offering new hope for patients with cancer, autoimmune conditions, and genetic disorders. However, the development and manufacturing of these therapies demand reagents of uncompromising quality, consistency, and safety. Qkine’s new Cell Therapy Grade proteins address this critical need by delivering:
- Defined manufacturing standards – Produced under controlled conditions with full traceability and regulatory support documentation
- Enhanced quality attributes – High purity, animal origin-free, low endotoxin levels, and reduced host cell protein/DNA contamination
- Scalability for clinical workflows – Consistent performance from research-grade to therapy-grade materials, enabling seamless transition to GMP production
- Regulatory readiness – Comprehensive Certificates of Analysis (CoA), Safety Data Sheets (SDS), and technical dossiers to support IND and clinical submissions
The portfolio supports a wide range of applications, including CAR-T and TCR therapies, NK cell expansion, iPSC differentiation, and ex vivo gene editing workflows.
In the first phase 14 proteins have been released, but all our current research use portfolio is available for CTG manufacturing on request, contact us for more information ryan.weber@matriqx.com.
For more information on our new cell therapy grade range:
Learn more about our animal origin-free cell therapy grade manufacture
Did you miss?
Why are Qkine’s new products labelled cell therapy grade and not GMP grade?
Qkine have recently released a range of growth factors and cytokines specifically designed to be suitable for cell and gene therapy manufacturing. Most recombinant protein manufacturers would describe these as 'GMP grade' but we haven't, so why are the products labelled cell therapy grade and not GMP grade?
Our latest blog explains what exactly is GMP and how our cell therapy grade proteins comply with GMP and ISO standards.
Animal origin-free protein manufacture for cell therapy
We have previously discussed the issues surrounding animal-derived products for research and why animal origin-free (AOF) alternatives are preferred. These include the potential for cross-contamination with other bioactive proteins or contaminants, which can introduce unwanted variability. When considering proteins used for the development of cell therapy, further assurance of purity and consistency is paramount, particularly due to the potential risk of harm through the application of an improperly manufactured product.
What's new?
Recombinant human activin A protein (Qk001-CTG)
Activin A is a TGF-β family growth factor frequently used to maintain pluripotency in induced pluripotent and embryonic stem cell cultures. It is essential for many stem cell differentiation protocols, including endoderm lineage differentiation and further maturation into hepatocyte and pancreatic cells for regenerative cell therapy applications.
Cell therapy grade human activin A protein is a high-purity mature bioactive dimer of 26 kDa. It is animal origin-free (AOF), carrier protein-free, and tag-free to ensure its purity with exceptional lot-to-lot consistency.
Qkine cell therapy grade high purity animal origin-free proteins are manufactured as GMP grade equivalents in an ISO 9001:2015-certified facility, under ISO 20399:2022 standards with GMP compliance, defined quality criteria and documentation.
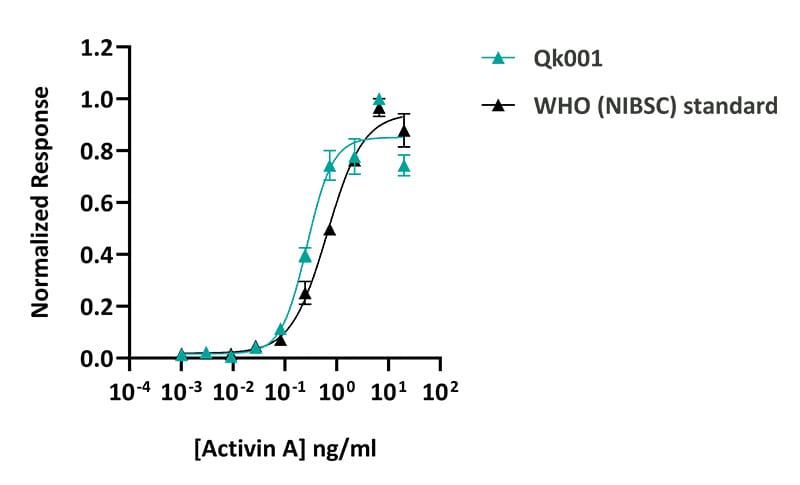
Qkine activin A was more active than the WHO (NIBSC) standard activin A. Activin A activity was determined using a CAGA luciferase reporter assay in transiently transfected HEK293 cells. Transfected cells were treated in triplicate with a serial dilution of activin A for 6 hours. Firefly activity was measured and normalized to the control Renilla luciferase activity. Qk001 #204564 EC50 = 0.27 ng/ml (10.4 pM). WHO (NIBSC) standard EC50 = 0.66 ng/ml.
Recombinant human IL-34 PLUS™ protein (Qk091)
Qkine have developed the only commercially available bioactive animal origin-free IL-34!
Qkine have optimized the sequence and manufacture of IL-34 PLUS™ to produce a highly and consistently bioactive protein (patent pending GB 2514413.0). Qk091 has a molecular weight of 25 kDa as a monomer and is supplied as a non-covalent dimer, highly pure, animal origin-free, tag free and carrier free for reproducible microglial differentiation and immune cell culture.
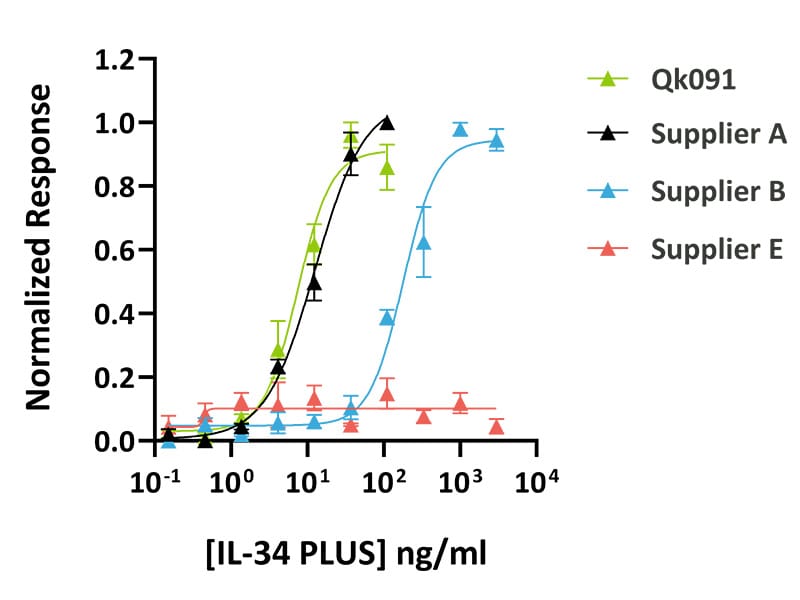
Qkine animal origin-free IL-34 PLUS™ was as bioactive as a mammalian cell-expressed IL-34 from an alternative supplier. IL-34 activity was determined using an SRE reporter assay in HEK293 cells co-transfected with the CSF1R receptor. Transfected HEK293 cells were treated in triplicate with a serial dilution of Qkine IL-34 PLUS™ (Qk091, green), mammalian cell-expressed IL-34 (Supplier A, black) and (Supplier B, blue) or bacterially-expressed IL-34 (Supplier E, pink) for 3 hours. Firefly activity was measured and normalized to the control Renilla luciferase activity.
Technotes
At Qkine data transparency is our promise. We test all new products against others on the market to ensure each is as bioactive or more, only then will they be approved for sale.
Each new lot is tested to ensure the bioactivity is consistent lot-to-lot, to ensure the most consistent cell cultures and assays. All data is available for all lots, and many are available as technotes.
Why not see if data on your protein of interest is available (if not ask!)
Technote | TPO (Qk098) bioactivity
Quantitative luciferase reporter assay shows Qkine TPO was more bioactive than an alternative supplier protein. Recombinant TPO activity was determined using a Promega serum response element luciferase reporter assay in HEK293T cells co-transfected with TPOR. Cells were treated in triplicate with a serial dilution of Qkine TPO (Qk098, green) or Peprotech TPO (Supplier B, black) for 3 hours. Firefly activity was measured and normalized to the control Renilla luciferase activity. Data from Qk098 lot #204752.
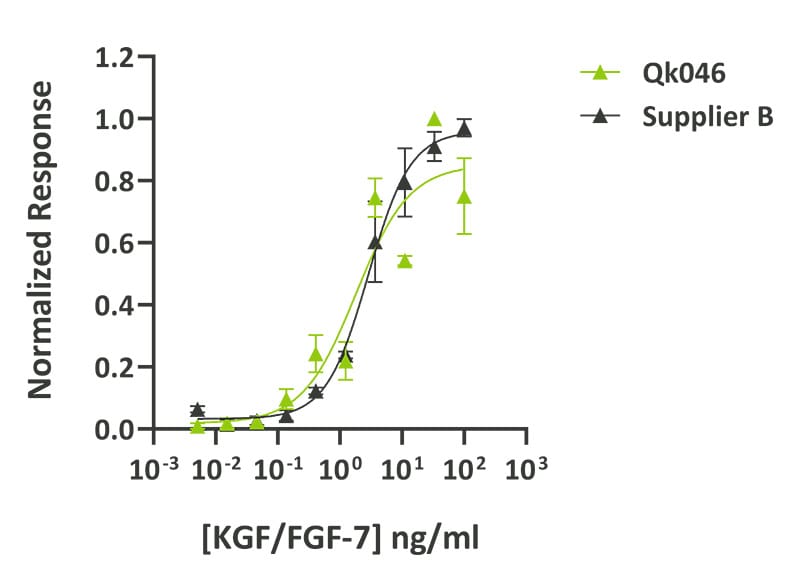
Technote | KGF/FGF-7 (Qk046)

Qkine KGF was as bioactive as KGF from an alternative major supplier. Bioactivity was determined using the Promega serum response element (SRE) luciferase reporter assay in transfected MCF-7 cells. Cells were treated in triplicate with a serial dilution of Qkine KGF (Qk046, green) or Peprotech KGF (100-19, Supplier B, black) for 4 hours. Firefly luciferase activity was measured and normalized to control Renilla luciferase activity. Data from Qk046 lot #104425.
Recent publications
Cardiomyocyte-expressed TGFβ signals to fibroblasts to program early heart maturation and adult myocyte identity
Minerath, RA et al. bioRxiv, September 2025
Researchers results indicate that TGFβ generated by cardiomyocytes in the embryonic heart mediates fibroblast differentiation that co-evolves the ECM environment that in turn programs cardiomyocyte maturation to establish their identity.
Used Qkine human TGF-β1 (Qk010)
LRRC58 defines an E3 ubiquitin ligase complex sensitive to cysteine abundance
All our growth factors are manufactured within a stringent quality framework, ensuring high-quality proteins that maintain robust, reproducible, and physiologically relevant stem cell and organoid cultures that adhere to our Nine-point Quality Commitment.
Upcoming Events
- Advanced Therapies USA 2025 | 18-19 November 2025 | Philadelphia, USA
- Alternative Protein 4.0 | 21/28 November 2025 | Online Event
- 3rd International Conference of CellAgri Portugal | 27-28 November 2025 | Olhão, Portugal
- ATMP Sweden 2025 | 27-28 November 2025 | Gothenburg, Sweden
- Mercia Stem Cell Alliance | 3 December 2025 | Keele, UK
- WORD+25 Research Celebration | 4 December 2025 | London, UK
- Assembloids & Complex Cell-Cell Interactions across Tissues & Organs | 8-11 December 2025 | CHSL, New York, USA
- ISSCR Boston International Symposium | 11-12 December 2025 | Cambridge, MA, USA
We'd always love to meet you at any of events, contact ryan.weber@matriqx.com to make an appointment.
What’s next?
Qkine are committed to producing the most high quality and bioactive growth factors and cytokines, coming soon to our extensive portfolio:
- Recombinant human activin B protein (Qk024) - Expected November 2025
- Recombinant human activin C protein (Qk026) - Expected November 2025
If you wish to be notified when proteins are released, contact us ryan.weber@matriqx.com

